 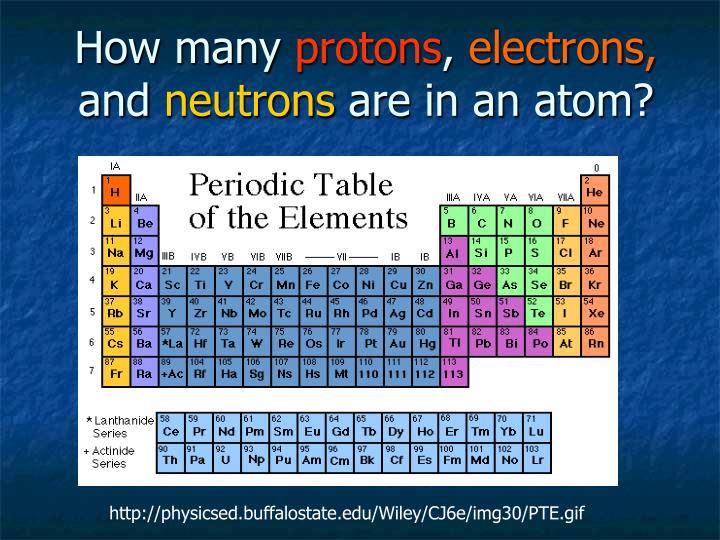
– Read “Atomic Weights: No Longer Constants of Nature”, Chem Int 33(2), 10–15 (2011), IUPAC Periodic Table of the Elements and Isotopes (IPTEI) for the Educational Community Coplen, Peter Mahaffy, Ian Mills, Roberto Marquardt, and more. Leigh, Sigurd Hofmann, Eric Scerri, Juris Meija, Norman E. See PAC (AOP ) for full details or visit Commission II.1 ĭownload the PDF version (letter size or A4) or A3 (PDF) or see earlier versionsĬheck out SPECIAL Chem Int Jan 2019 - International Year of the Periodic Table (IYPT) - with contributions by Jan Reedijk, Natalia Tarasova, G.J. For elements that lack isotopes with a characteristic isotopic abundance in natural terrestrial samples, the mass number of the nuclide with the longest confirmed half-life is listed between square brackets. It does not store any personal data.The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights ( CIAAW), compiled as part of the 2021 Table of Standard Atomic Weights 2021. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. There are three stable isotopes of the element, with atoms having 10 neutrons (neon-20), 11 neutrons (neon-21), and 12 neutrons (neon-22). The number of protons is equal to the number of electrons, unless there’s an ion superscript listed after the element.Įach neon atom has 10 protons. That number is equal to the number of protons. The easiest way to find the number of protons, neutrons, and electrons for an element is to look at the element’s atomic number on the periodic table. How do I know how many electrons and protons an element has? Square the ring number, then multiply the result by two. Neon on the periodic table has an atomic number of 10, so it has 10 electrons. For example, assume you want to know how many rings are in the element neon. The number of electrons is equal to the atom ‘s atomic number, which is at the top left of the element. How do you know how many electrons an element has? To be an element, a substance has to at least have protons, since these particles define the type of element. 
Since the atomic number of calcium is 20 and the charge is positive, this means the ion has 20 – 2 or 18 electrons. Since the atom is electrically neutral, the number of electrons must equal the number of protons. The number of protons also determines the identity of the element. Protons contribute to the mass of an atom and provide the positive charge to the nucleus. Why do we use protons to identify an element? The number of neutrons is equal to the difference between the mass number of the atom (M) and the atomic number (Z). The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus. The number of electrons in a neutral atom is equal to the number of protons. What is defined by its number of electrons? The number of protons determines an element’s atomic number and is used to distinguish one element from another. Neutral atoms of each element contain an equal number of protons and electrons. What is an element is determined by the number of? The atomic number gives the “identity” of an element as well as its location on the periodic table. It also tells you the number of electrons in a neutral atom of that element. The number of protons in one atom of an element determines the atom’s _, and the number of electrons determines the Charge of the element. What does the number of electrons determine in an element? 4 How do I know how many electrons and protons an element has?.2 What is defined by its number of electrons?.1 What does the number of electrons determine in an element?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
